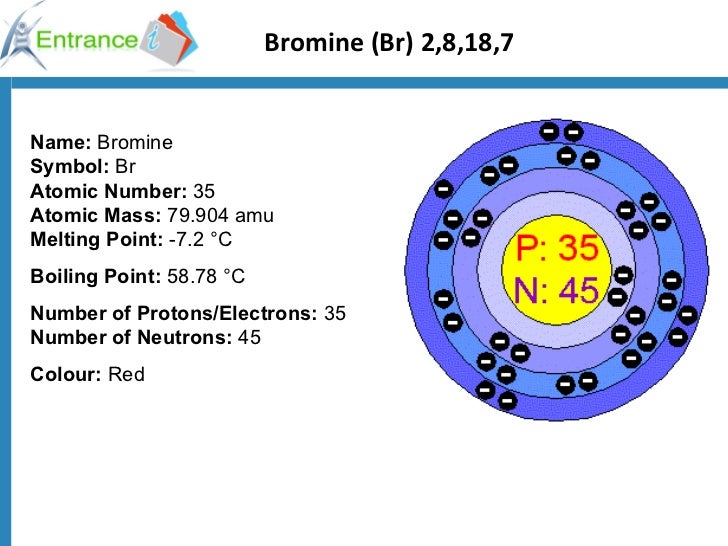 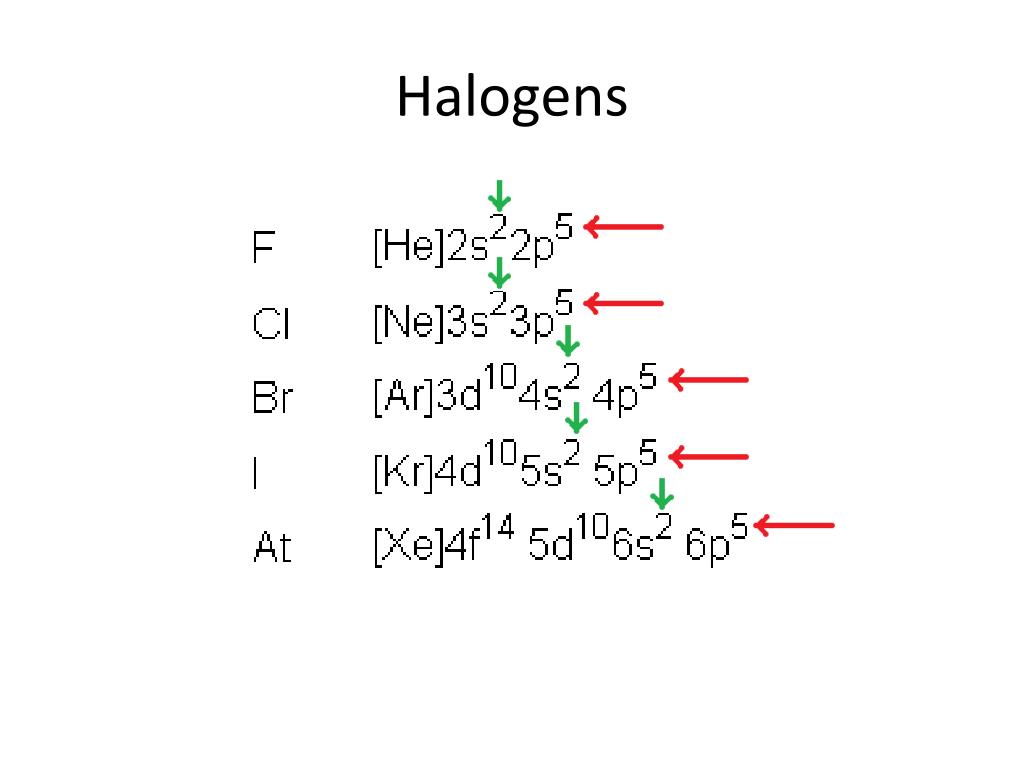
The Schrödinger equation is similar in form to equations for the propagation of waves, which is why originally quantum mechanics was called wave mechanics, but there are significant differences between quantum wave functions and those that describe real waves. We focus on the properties of the wave functions that are the solutions of Schrödinger’s equations. He then worked at Princeton University in the United States but eventually moved to the Institute for Advanced Studies in Dublin, Ireland, where he remained until his retirement in 1955.Īlthough quantum mechanics uses sophisticated mathematics, you do not need to understand the mathematical details to follow our discussion of its general conclusions. When Hitler came to power in Germany, Schrödinger escaped to Italy. He was notorious for his intense dislike of memorizing data and learning from books. Schrödinger’s unconventional approach to atomic theory was typical of his unconventional approach to life. It successfully describes the energies and spatial distributions of electrons in atoms and molecules. In doing so, Schrödinger's theory today is described as quantum mechanics A theory developed by Erwin Schrödinger that describes the energies and spatial distributions of electrons in atoms and molecules. In 1926, an Austrian physicist, Erwin Schrödinger (1887–1961 Nobel Prize in Physics, 1933), developed wave mechanics, a mathematical technique that describes the relationship between the motion of a particle that exhibits wavelike properties (such as an electron) and its allowed energies. Scientists needed a new approach that took the wave behavior of the electron into account. The paradox described by Heisenberg’s uncertainty principle and the wavelike nature of subatomic particles such as the electron made it impossible to use the equations of classical physics to describe the motion of electrons in atoms. To apply the results of quantum mechanics to chemistry.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
